|
Home |
Consulting |
Training |
Expert Witness |
Failure Analysis |
Design Review |
Corrosion Test |
Corrosion Software
|
Coatings
|
Materials Selection |
Cathodic Protection |
>>> |
|
 Corrosion Modeling Software and Corrosion Prediction Software Series CIPAL-Compass®: Copper-Induced Pitting in Aluminium Alloys - Modeling, Life Prediction, and Process Control Version 9.23
Anytime Anywhere Any Device Any OS
No USB dongles No
installation
No Browser Plug-ins
|
|
Why WebCorr |
Performance Guarantee |
Unparalleled Functionality |
Unmatched Usability |
Any Device Any OS |
Free Training
& Support |
CorrCompass |
|
|
Features and Component of CIPAL-Compass |
|
|
CIPAL-Compass is the only device and OS independent software on the market for the modeling and life prediction of copper-induced pitting in aluminium alloys used in the microelectronics and semiconductor industry, the chemical process industry, the power generation industry, the water treatment industry, and any industry/sector where aluminium alloys come into contact with waters or process fluids that contain trace amount of copper ions. Designers, engineers, architects, consultants, maintenance and inspection personnel can quickly assess and quantify the impact of copper ions on the pitting corrosion of aluminum alloys, the pit depth, the pitting rate, the remaining life of their components or systems, and the critical level of copper ions for process control. CIPAL-Compass utilizes machine learning and cloud computing and works anytime, anywhere, on any device running any OS without the need to install or download anything.
CIPAL-Compass consists of two modules:
Overview and Application Examples of CIPAL-Compass for Modeling and Life Prediction of Aluminum Alloys
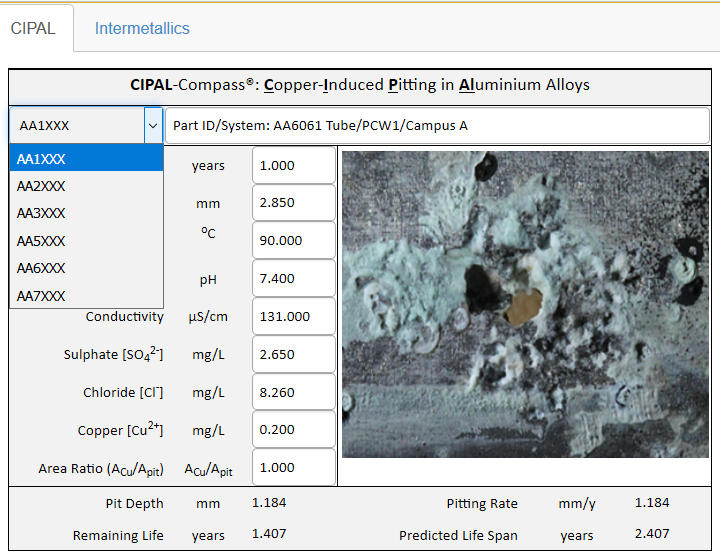
Figures below show the screen shots of CIPAL-Compass. CIPAL-Compass quantitatively determines the effect of the chemistry of water or process fluid on the pitting corrosion of aluminum alloys: temperature, pH, conductivity, sulphate, chloride, and copper ions. The outputs from CIPAL-Compass includes the pit depth, the pitting rate, the predicted life span, and the remaining life of the component.
Figure 1 Overview of CIPAL-Compass
Figure 2 Selection of Aluminium Alloys Series in CIPAL-Compass
Users of CIPAL-Compass start by selecting the aluminum alloy series from the dropdown list. The specific grade of aluminum alloy in a particular series is not required as all grades within the same series showed similar corrosion behaviour.
After selecting the aluminium alloys series, the next step is to enter the chemistry parameters of the water or process fluid. Temperature, pH, conductivity and concentrations of sulphate, chloride, and copper ions are all critical factors that influences both the initiation of pitting and the propagation rate of pitting in aluminum alloys.
CIPAL-Compass also models the galvanic effect of copper deposition on the aluminium surface by taking into account of the surface area ratio of the copper deposit to the pit opening.
Service Life Prediction for Process Piping in Semiconductor Manufacturing In semiconductor manufacturing, the process cooling water is frequently contaminated with copper ions, which will deposit on aluminium alloy AA6061 piping surface and induce pitting corrosion in aluminum alloy AA6061. The temperature of process cooling water is about 90°C and the pipe wall thickness is 2.85 mm. The chemistry of the process water is as entered. CIPAL-Compass predicts that the piping would leak in about 2.5 years after operation due to copper-induced pitting in the aluminum alloy.
Figure 3 Service Life Prediction for Process Piping in Semiconductor Manufacturing
Overview of CIPAL-Compass for Intermetallics-Induced Pitting in Aluminum Alloys Intermetallics, their size and distribution in aluminium alloys, are critical factors influencing the alloys' mechanical strength and corrosion resistance properties. Some intermetallics are anodic to the aluminum matrix while others are cathodic to the matrix. The anodic intermetallics will corrode preferentially, creating cavities/holes in the aluminium matrix (Figure 4). The cathodic intermetallics will not corrode themselves but will cause corrosion of the matrix around their perimeters (Figure 5). Figures below show overviews of the CIPAL-Compass module for Intermetallics-Induced Pitting in Aluminum Alloys.
Figure 4 Overview of CIPAL-Compass for Intermetallics-Induced Pitting in Aluminum Alloys
Figure 5 Overview of CIPAL-Compass for Intermetallics-Induced Pitting in Aluminium Alloys
A dozen of intermetallics commonly found in aluminium alloys are available for selection in CIPAL-Compass (Figure 6). If you cannot find the intermetallics of your interest in the list, do let us know through the Contact Us link and we will conduct the necessary tests to generate the required data for inclusion in the software.
Figure 6 Common Intermetallics in Aluminium Alloys in CIPAL-Compass
Users simply enter the conductivity and the operating temperature of the process water/fluids, CIPAL-Compass takes care of the rest.
Outputs from the Intermetallics module of CIPAL-Compass includes the following:
WebCorr can customize CIPAL-Compass for your specific process fluids and alloys used in any industry from general engineering to wafer fabrication. Click here to contact us for licensing details and experience the power of CIPAL-Compass. |
|
|
CIPAL-Compass, giving you the right directions in Pitting Prediction in Aluminum Alloys |
|
|
Home | Contact Us | PDF |
Copyright © 1995-2024. WebCorr Corrosion Consulting Services. All rights reserved. |